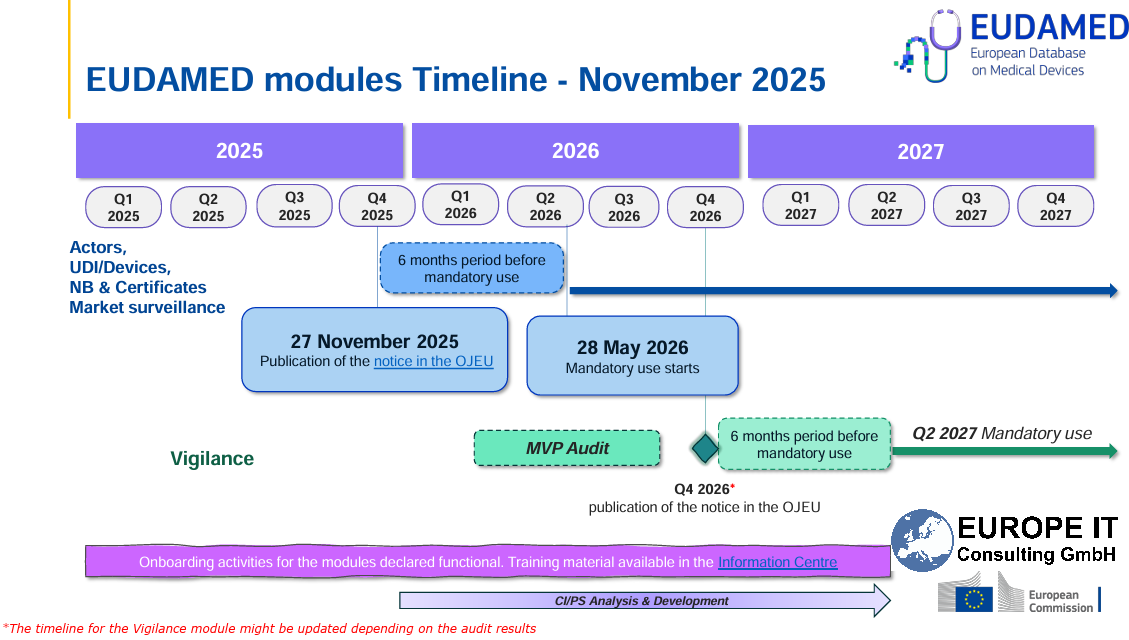
Current Compliance Deadlines
Stay compliant: The mandatory use of EUDAMED (EU) for medical device registration is approaching. Note the current transition periods:
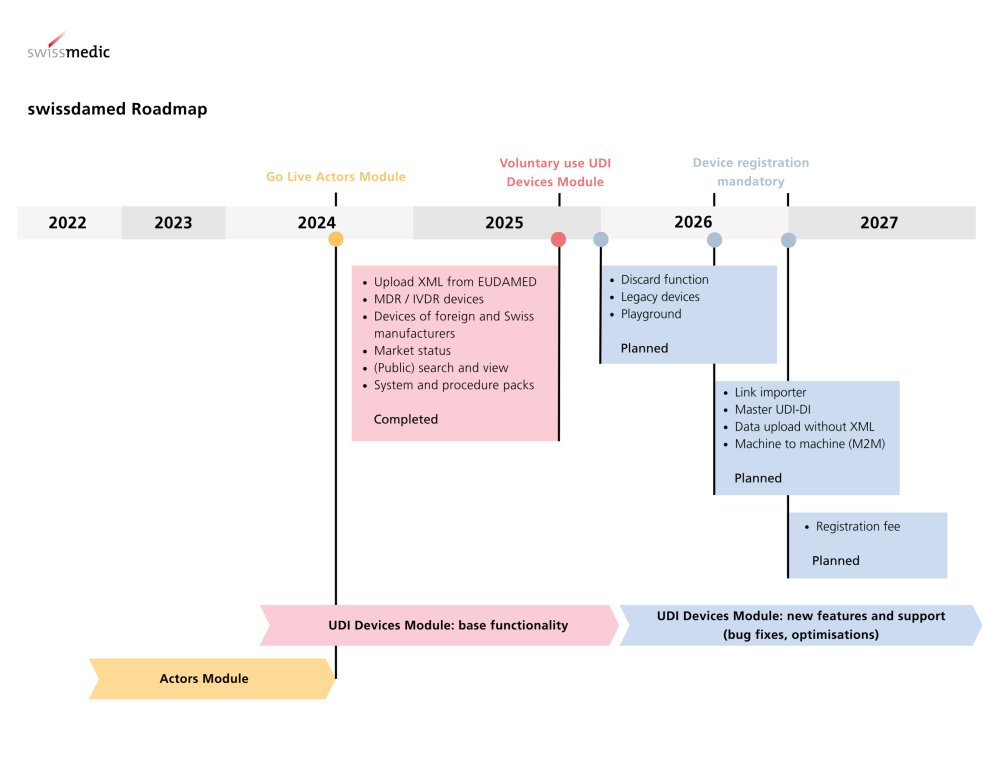
In Switzerland, registration via swissdamed (CH) is also already mandatory for many actors. Parallel requirements require a synchronous strategy:
We support you in safely meeting both deadlines through automated processes and our combined GSP solution.